Heat-stable carbetocin has been added to the WHO Essential Medicines List for the prevention of excessive bleeding after childbirth
- Heat-stable carbetocin has been added to the WHO Essential Medicines List of uterotonics for the prevention of excessive bleeding after childbirth 1
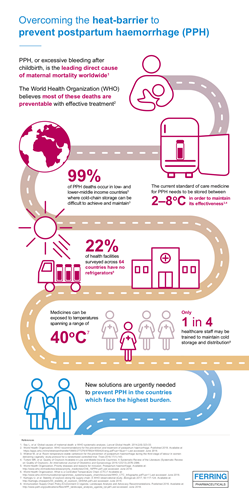
- Excessive bleeding after birth, also known as postpartum haemorrhage (PPH), is the leading direct cause of maternal mortality worldwide 2
- WHO guidelines recommend heat-stable carbetocin for PPH prevention in settings where oxytocin is unavailable or its quality cannot be guaranteed, and where its cost is comparable to other effective uterotonics 3*
- Heat-stable carbetocin was developed by Ferring Pharmaceuticals to address limitations in refrigeration and cold-chain transport of medicines in low- and lower-middle income countries 4
Saint-Prex, Switzerland – Tuesday, 09 July 2019 –
Ferring Pharmaceuticalstoday welcomes the World Health Organization’s (WHO) addition of heat-stable carbetocin to the WHO Model List of Essential Medicines (EML) of uterotonics for the prevention of excessive bleeding after birth.1 The EML identifies medicines the WHO deems essential for addressing the most important public health needs globally.

Nepal: Dinesh felt helpless as his wife Tulasi was rushed to have emergency blood transfusions after experiencing postpartum haemorrhaging (PPH) following the birth of their first child. His mind raced, imagining raising their new born on his own and without a mother. Tulasi recovered and Dinesh is thankful for this every day.
Every year, 14 million women are affected by excessive bleeding after birth, also known as postpartum haemorrhage (PPH).5 Although most deaths are preventable,6 PPH is the leading direct cause of maternal death worldwide,2 causing approximately 70,000 deaths per year,2 99% of which occur in low- and lower-middle income countries.5
“The WHO’s addition of heat-stable carbetocin to the Essential Medicines List brings us one step closer to tackling this life-threatening condition that impacts thousands of women and their families,” said Klaus Dugi, Chief Medical Officer, Ferring Pharmaceuticals. “This milestone is thanks to collaboration from stakeholders around the world, and demonstrates how work across both the public and private sectors can help achieve development goals in global health. As the manufacturer, Ferring is now seeking registrations for heat-stable carbetocin and, once approved, our priority is to make heat-stable carbetocin available at an affordable and sustainable price** in public sector healthcare facilities in low- and lower-middle income countries where cold-chain transport and refrigeration of medicines is often difficult to achieve or maintain.”
This addition to the EML follows the 2018 update to the WHO’s recommendations on uterotonics for the prevention of PPH.3 The guidelines recommend heat-stable carbetocin for the prevention of excessive bleeding after all births in settings where oxytocin is unavailable or its quality cannot be guaranteed, and where its cost is comparable to other effective uterotonics.3 The CHAMPION trial, the largest clinical trial in PPH prevention, showed that heat-stable carbetocin is non-inferior to current standard of care oxytocin, for the primary outcome of ≥500 ml blood loss or additional uterotonic use, after vaginal birth.7 Heat-stable carbetocin remains effective at high temperatures,8 addressing a significant limitation of oxytocin, which must be stored and transported at 2 – 8°C.9, 10
Carbetocin is currently registered in more than 80 countries worldwide for the prevention of PPH due to uterine atony following caesarean section.11 Ferring is now seeking registrations for heat-stable carbetocin for the prevention of PPH following all births. Once approved, the heat-stable formulation of carbetocin will be available at an affordable and sustainable price** to publicly controlled or publicly funded healthcare facilities and healthcare facilities operating on a social marketing basis in low- and lower-middle income countries.
About the EML
The WHO Model List of Essential Medicines (EML) serves as a guide for the development of national and institutional essential medicine lists and is updated and revised every two years by the WHO Expert Committee on Selection and Use of Medicines.
About the CHAMPION trial
CHAMPION (Carbetocin Haemorrhage Prevention), the largest clinical trial in the prevention of PPH, was a double-blind, randomised, non-inferiority trial designed to compare the effectiveness and safety of investigational heat-stable carbetocin to oxytocin in the prevention of PPH after vaginal birth. The trial, funded by MSD for Mothers*** and conducted by the WHO Department of Reproductive Health and Research including the UNDP-UNFPA-UNICEF-WHO-World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP), enrolled nearly 30,000 women in ten countries including Argentina, Egypt, India, Kenya, Nigeria, Singapore, South Africa, Thailand, Uganda and the UK. Heat-stable carbetocin is a long-acting uterotonic developed by Ferring Pharmaceuticals. The results demonstrated that heat-stable carbetocin is non-inferior to the standard of care, oxytocin, for the primary outcome of > 500 ml blood loss or additional uterotonic use. Non-inferiority was not demonstrated for the secondary primary outcome of blood loss of 1000 ml or more, however, the trial was underpowered for this outcome. There were no significant differences between groups in other measures of bleeding or in adverse effects.
* The availability of heat-stable carbetocin is subject to regulatory review and approval in relevant countries.
** This price is a subsidised price of $0.31 +/- 10% per ampoule of 100 µg heat-stable carbetocin. This is comparable to the current United Nations Population Fund price for oxytocin of $0.33 per unit (10 I.U.).12
*** MSD for Mothers is an initiative of Merck & Co., Inc., Kenilworth, N.J., U.S.A.
About Ferring Pharmaceuticals
Ferring Pharmaceuticals is a research-driven, speciality biopharmaceutical group committed to helping people around the world build families and live better lives. Headquartered in Saint-Prex, Switzerland, Ferring is a leader in reproductive medicine and women’s health, and in speciality areas within gastroenterology and urology. Ferring has been developing treatments for mothers and babies for over 50 years and has a portfolio covering treatments from conception to birth. Founded in 1950, privately-owned Ferring now employs approximately 6,500 people worldwide, has its own operating subsidiaries in nearly 60 countries and markets its products in 110 countries.
Learn more at www.ferring.com, or connect with us on Twitter, Facebook, Instagram, LinkedIn and YouTube.
For more information, please contact
Bhavin Vaid
Head of Corporate Communications
+41 58 301 0952 (direct)
+41 79 191 0632 (mobile)
bhavin.vaid@ferring.com
Lindsey Rodger
Senior Manager, Corporate Communications
+41 58 451 4023 (direct)
+41 79 191 0486 (mobile)
lindsey.rodger@ferring.com
References
- WHO Model List of Essential Medicines. Available at: https://apps.who.int/iris/handle/10665/325771 Last accessed: July 2019
- Say L, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Global Health. 2014; 2(6):e323-e333. Available at:
https://doi.org/10.1016/S2214-109X(14)70227-X Last accessed: July 2019 - WHO. WHO recommendations: Uterotonics for the prevention of postpartum haemorrhage. Available at: https://apps.who.int/iris/bitstream/handle/10665/277276/9789241550420-eng.pdf?ua=1 Last accessed: July 2019
- World Bank. World Bank Country and Lending Groups. Published 2019. Available at: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups Last accessed: July 2019
- WHO. Priority diseases and reasons for inclusion. Postpartum haemorrhage. Available at: http://www.who.int/medicines/areas/priority_medicines/Ch6_16PPH.pdf Last accessed: July 2019.
- WHO. Maternal mortality – Fact sheets. Published 2018. Available at: www.who.int/news-room/fact-sheets/detail/maternal-mortality Last accessed: July 2019
- Widmer M, et al. Heat stable carbetocin vs oxytocin to prevent hemorrhage after vaginal birth. New England Journal of Medicine 2018;379:743-752
- Malm M, et al. Development and stability of a heat-stable formulation of carbetocin for the prevention of postpartum haemorrhage for use in low and middle-income countries. Journal of Peptide Science 2018;e3082
- Widmer M, et al. Room temperature stable carbetocin for the prevention of postpartum haemorrhage during the third stage of labour in women delivering vaginally: study protocol for a randomized controlled trial. Trials 2016;17(1):143
- Torloni MR, et al. Quality of Oxytocin Available in Low and Middle-Income Coun
- Ferring Pharmaceuticals. Data on file. 2019
- UNFPA Procurement Services. Oxytocin 10 I.U./ml injection in 1ml ampoule. www.unfpaprocurement.org/products?id=OXYTOCIN_10IU/ML Last accessed: July 2019